As a manufacturer of orthopaedic medical devices under the QS Regulation and ISO 13485, your company is responsible for the implementation and maintenance of a formal Quality Management System (QMS).
When a nonconformity is identified, your company will determine the significance, associated risk and potential for recurrence. You may then decide that the nonconformity has little associated risk or is unlikely to recur. In such cases, your Review Board may decide only to carry out a correction (rather than a full blown, costly and time-consuming corrective action).
Your Quality Review Board (QRB) may encounter issues or trends from quality data that have not actually caused a nonconformity, but may do so in the future. Such situations may call for preventive action.
Definitions
- Correction: An action to eliminate a detected nonconformity. A correction can be made in conjunction with corrective action. Corrections can be, for example, re-work or retraining.
- Corrective action (CA): An action to eliminate the cause of a detected, systemic nonconformity or other undesirable situation. There can be more than one cause for nonconformity. Corrective action is taken to prevent recurrence, whereas preventive action is taken to prevent occurrence. The CAPA system is always described as an integral part of the QMS and is driven by Planning, Measurement and Analysis, Improvement and Management Support.
- Preventive action (PA): Action to eliminate the cause of a potential nonconformity or other undesirable situation. Preventive action is taken to prevent occurrence, whereas corrective action is taken to prevent recurrence.
- Nonconformity: Non-fulfillment of a requirement.
- Quality Review Board (QRB): A cross-functional group of process owners and ad hoc representatives deemed by a company to be responsible for deciding the disposition of all defective products and out-of-compliance processes (nonconformances).
Management Buy-in
It’s critical to establish the involvement of management during, for example, Management Review or during QRB sessions in actions taken in response to nonconformities or potential nonconformities. Management is responsible for improvement, compliance and enablement of the QMS at all levels of your company.
In the process of planning measurement and analysis, your QRB needs to take into account data sources, the measurement of the data elements within each data source, the frequency of monitoring and the analysis to be performed within a data source, or across data sources. Managing risk is a primary driver for making these determinations and giving disposition to nonconformities in a compliant manner. The measurement of data elements should be done in a way that ensures that your company processes are effective in managing the operations and maintaining an operative, process-based QMS. Each of the data elements on your company’s dashboard should be established, with specific requirements for measurements that are monitored routinely.
The Quality Review Board: A Risk-based, Cross-functional Team of Process Owners
The QRB consists of at least these representatives from the following processes within your QMS:
- Functional process owners responsible for the viability of second level procedures.
- Suppliers, contractors and consultants who could advise the QRB, e.g. requiring an expert, brought to the QRB on an ad hoc basis.
- Any member of the company who could advise the QRB on similar subjects.
The QRB meets when there is a necessity to resolve or redirect issues of alleged, higher risk subjects. Meetings can be held on a revolving and formal schedule as well, as long as the entire proceedings are documented and aligned with the strategic objectives of your company. Management is always apprised of the activities of the QRB in a timely and definitive manner and, more often than not, the minutes and trends emanating from these sessions can be inputs to formal Management Review. The Process Owner for Corrective and Preventive Action (CAPA) usually conducts and facilitates all QRB Meetings and makes arrangements to organize all objective evidence originating for, from or because of these decision-making sessions. Management should be involved in a review and confirmation that actions taken were effective and did not introduce new issues or concerns. The following questions could be considered at appropriate times throughout the process and be revisited during ensuing reviews:
• Has the problem been comprehensively identified?
• Has the extent of the problem been identified (e.g. range of affected devices, patient outcome, process, production lines, operator)?
• Have the root cause/contributing factors of the problem been identified and addressed?
• Has the improvement action been defined, planned, documented, verified and implemented?
Risk management practices will play a big part in every decision made by the QRB, and these decisions must be traceable back to a rationale for risk-based conclusions, e.g. using FMEA (product and/or process Failure Mode Effects Analysis) results toward mitigated resolutions and outputs. All potential changes to product or process must be first judged in terms of design controls, i.e. re-design, design change, etc. If a re-design is considered a viable option, a documented and then approved, risk-based rationale must be issued to decide upon the final decision.
Examples of activities that could be addressed by the QRB:
• Individual occurrences and recurrences with alleged high risk
• Trended occurrences and recurrences
• Internal and supplier CAPAs
• Needs-assessment concerning the QMS and supplier management activities
• Purchasing strategies
• Non-conforming product
• Internal and supplier audit findings
• Complaints
• Service reports
• Quality records
• Documents and procedures
• Improvement opportunities
• Customer feedback
• Outstanding actions from previous QRB meetings
• Revised regulatory requirements
• Returned products
• Regulatory activities (e.g. recalls, FDA 483, warning letters, re-submissions of pre-market notifications, addendums to pre-market approvals, etc.)
• Sources of quality data (e.g. post-commercialization trends, adverse events that could represent the filing of an MDR and/or MDV, possible re-design opportunities, etc.)
• Remedial actions for calibration
• The master validation plans for software and process
• Risk management adjustments
• Resource allocation
• Supplier report cards
• Human factors (that could lead back into the design controls area) for devices
• Quality plans and planning
• Decisions concerning the lifetime of the medical device
Risk Analysis Process and Approach
A risk analysis should be performed and documented for each device using the most appropriate method for your company’s specific requirements. The use of relevant information from similar devices and processes as a starting point can be advantageous and will continue to grow as a data platform is realized. This starting point is directly related to the type of orthopaedic devices that you are manufacturing, the complexity of your QMS and the severity/occurrence demonstrated.
The documentation of the risk analysis shall include the following inputs: 1) description of the process within the product or process lifecycle, 2) identification of the person performing the analysis, 3) scope and date of the analysis and 4) intended use/purpose and identification of characteristics related to process and products. For each identified hazard situation, a decision must be made, using the criteria in the risk management plan, whether the estimated risk is so low that risk reduction need not be pursued. The risk analysis shall ensure that the risks from all identified hazard situations are considered during QRB discussions, during design reviews and management review sessions, as well.
The following presents a sample of agenda and topics addressed at a typical QRB meeting.
• Premise and Scope – QRB Rationale for holding this meeting
• Cross-Functional Team comprising these process owners (core team and on an as-needed/ad hoc basis)
• Core Team by Function(s)
• Timely Subjects (related to risk)
• Recommended Training
• Recommended CAPA
• Outputs of a typical QRB Meeting (deliverables)
• Participants (sign/date)
Conclusions
It is the intention of the QRB that continual improvement to the QMS shall occur regularly and frequently, but not necessarily without interruption. These activities should be documented and facilitated via the Process Owners at your company.
Applying the principle of system approach to management typically leads to:
• Structuring a system to achieve the organization’s objectives in the most effective and efficient way.
• Understanding the interdependencies among the processes of the QMS.
• Structured approaches that harmonize and integrate processes.
• Providing a better understanding of the roles and responsibilities necessary for achieving common objectives, and thereby reducing cross-functional barriers.
• Understanding organizational capabilities and establishing resource constraints prior to action…knowing your company’s limitations.
• Targeting and defining how specific activities within a system should operate.
• Continually improving the system through measurement and evaluation.
When opportunities for improvement are realized, they should be prioritized by focusing on the prospects that will produce the greatest return on investment for your orthopaedic manufacturing company. Not every problem or non-conformance requires corrective action. Whether corrective action is needed varies by type of device being manufactured, risks involved with each occurrence and the severity of the output. The decision may be left to process owners, as members of the QRB. In any case, it is not a decision for only one individual to make. FDA will emphasize that point throughout the inspection, and always place the ultimate responsibility for correcting problems upon top level management.
The following flowchart in Exhibit 1, supplied by the Global Harmonization Task Force, summarizes guidance on corrective action, preventive action and related QMS processes.
Exhibit 1: Processes for Measurement, Analysis and Improvement: Guidance on Corrective Action and Preventive Action/Related QMS Processes for Medical Devices
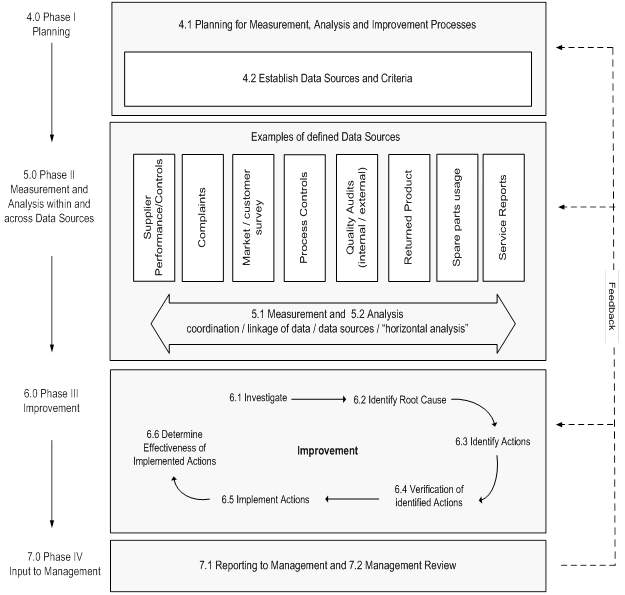
Source: Global Harmonization Task Force – GHTF/SG3/N18:2010
John Gagliardi has had success over the past 40 years in the Medical Device and Pharmaceutical industries because of his practical approach to process-orientation and business. He has been actively involved in research and development, quality assurance, training, operations, process architecture, FDA inspections and regulatory affairs. John specializes in building systems in a compliant and business-ready manner. Reach John via email.
MidWest Process Innovation, LLC
513-573-0085 (phone)
www.midwestprocessinnovation.com