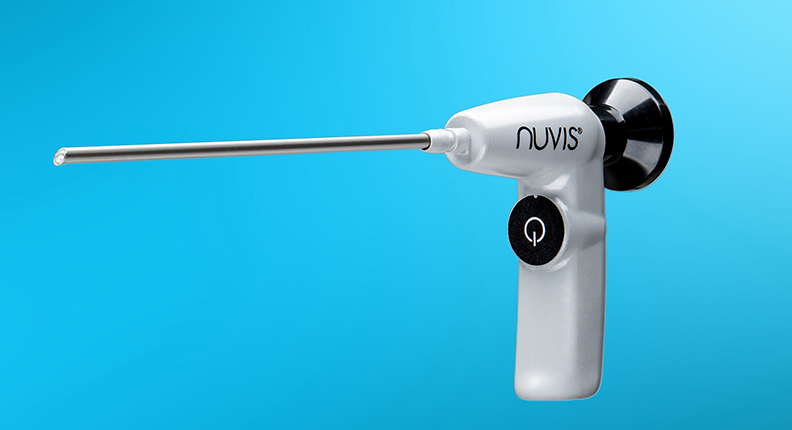
The orthopedic industry has embraced a variety of single-use devices, recognizing the benefits of reduced risk for infection and elimination of downtime during sterilization or repair. Integrated Endoscopy is taking O.R. efficiency to a new level with its NUVIS (pronounced NU-VEE) system, a single-use arthroscopic surgical solution.
Typically, single-use devices become viable when significant cost reduction can be offered without sacrificing performance of the reusable version. With that goal at the forefront, the NUVIS system has been optimized over the past eight years since its inception, with a significant redesign within the past three years to get it to its current iteration – a 4.0mm, cordless, battery-operated, single-use arthroscope.
“Our product is able to deliver the same high-definition (HD) optical quality as a reusable scope, and will support all current camera systems in the marketplace today,” said Rob Cripe, Chief Commercial Officer for Integrated Endoscopy. “We’ve reduced our cost to the point where the costs of maintaining, repairing and reprocessing a reusable device are largely neutral to using a single-use device like ours. Over the next several years, the rigid scope market is going to start converting at a higher rate, and eventually, we think that 100% of that marketplace will be converted to a single-use device.”
Innovative Performance
Currently, there are no other single-use HD arthroscopes for surgery on the market, though there are companies that are producing corded, single-use needle scopes for diagnostic purposes. Integrated Endoscopy is focused on the O.R.
“We’ve eliminated the cord that goes to the light source by embedding our own LED at the tip powered by a AA alkaline battery inside the handle of the scope,” Cripe said. “So, not only have we eliminated a piece of capital equipment and the heavy cord in the surgical field, but we’ve also delivered H.D. image quality and kept the cost low enough to allow for wide-scale adoption around the world.”
Traditional setups have a fiber light cord that connects to a large, hot light source on the arthroscopy tower. The cords are inconvenient for surgeons as they operate, have a risk of breaking, and they often can drop past the sterile field. Furthermore, the heat they give off can be a major safety factor. NUVIS eliminates the cords and doesn’t produce heat higher than 38℃.
The NUVIS system is adaptable and easily integrates into existing surgical equipment. It can be used on any camera in the marketplace that has a universal C-mount coupler, which is a device that connects the scope to the camera head and allows the image to be transmitted onto the monitor. The eyecup on the NUVIS scope is the same as traditional scopes, so there’s no need for a change in equipment.
“We’re coming in with a completely different value proposition, which is to address the efficiencies in the O.R. with a single-use scope that can be used along with your existing equipment,” Cripe said. “We’re not trying to displace all of the reusable arthroscopes today; we’re going in as the backup to your existing equipment, including arthroscopes, because downtime and breakage happen every day in every O.R.”
Design Challenges
The first major design challenge that Integrated Endoscopy faced with NUVIS was the ability to deliver H.D. image quality at the right price point.
“Our challenge was to develop a single-use scope that offered the same image quality as the current reusable for a cost that is low enough that the marketplace will adopt it,” Cripe said.
Integrated Endoscopy was able to deliver HD image quality while keeping costs down partly by reducing the number of glass lenses and spacers in the arrangement of optical components, called the optical train. The glass elements are essentially small lenses, and there are often more than 60 glass elements inside a reusable scope. Due to its innovative design, the NUVIS system significantly reduces the number of these elements while maintaining HD image quality.
“The actual design of the optical train was key to the reduction of glass elements and in some cases, we combined the glass pieces so that they can be made as one unit instead of multiple units,” Cripe said. “We also changed from what is known as molded glass technology to polished glass technology. Molded glass is a way of making a lens that turns out to be expensive and not as reliable. Borne out of cell phone technology, polished glass lenses now exist in a lot of different things, and so we did our homework to identify some suppliers that could get us the quality that we needed in polished glass.”
With the image quality at the right level, the next step was to eliminate the fiber optics, which they were able to do by putting an LED light at the tip of the scope.
“That was a big challenge as well, due to the lack of space inside the four-millimeter tube. You have very little room to work with when you’re placing other components inside the tip of the scope,” Cripe said. “The challenge of fitting a small enough light inside the tip, along with a reduced number of elements, was the biggest challenge to being able to produce a product that would still give the same performance at a cost that the market would bear.”
Once the design was locked in, the next hurdle was consistent manufacturing.
“We had to do a lot of work to make sure that we could manufacture every single scope with the same level of quality, from the design of the optical train and lenses themselves, to being able to check it along the way,” Cripe said. “That’s unique to the way that we build our scopes. Every time we place an element, we are able to check image quality, so you’re not checking it for the first time after it’s completed.”
Future Plans
The idea of a single-use scope spurred a vision for capital equipment in the form of an arthroscopy system that is based on wireless and battery-operated technology, which doesn’t currently exist in the arthroscopic space.
After Integrated Endoscopy started seeing a positive response from customers with the elimination of the light cord, they decided to explore getting rid of the cord on the camera head.
“Customers started telling us that, if you can address the capital budget here in an impactful way, you’ll have a really good chance of adopting more use of the single-use scope,” Cripe said.
The company is developing an arthroscopy tower system built on the concept of significantly reducing the cost of capital equipment. The system is expected to go to market sometime in 2021.
“Once we start selling the arthroscopy tower, we will then be in the marketplace with a full arthroscopy system, highly disruptive to the current state of affairs,” Cripe said. “By utilizing wireless and battery technologies, various pieces of capital equipment such as light sources, camera boxes, image management, printers and even shaver consoles can be eliminated in this new tower design, which in turn lowers costs. When we launch the tower system, it’s going to be a compelling conversation for the customer to see that the starting point for our full arthroscopy system will be 80%-90% less than the list pricing of existing equipment in the market today.”
The intent is to disrupt the marketplace in a way that addresses real issues that their customers face, starting with the arthroscope. While the scope is not yet fully launched in the market, Integrated Endoscopy has executed hundreds of evaluations worldwide and has sold a small amount of the product in various markets outside the U.S. while they ramp up their manufacturing capabilities. They are currently in their third round of financing.
The wireless, single-use NUVIS arthroscope is FDA 510(k) cleared, CE Marked and approved in Australia, India and other markets.
“The scope itself is essentially complete, and we sold small quantities at the beginning of the year,” Cripe said. “We are positioning NUVIS to be the backup to all your existing equipment. We can completely eliminate waiting on a scope for any reason and you can use this on your existing equipment; it’s immediately available, because it’s sterile, in a box and on the shelf. Your efficiencies improve and the costs associated with processing, repairs and downtime of reusable arthroscopes in today’s O.R. are essentially gone.”
HT
Heather Tunstall is a BONEZONE Contributor.




