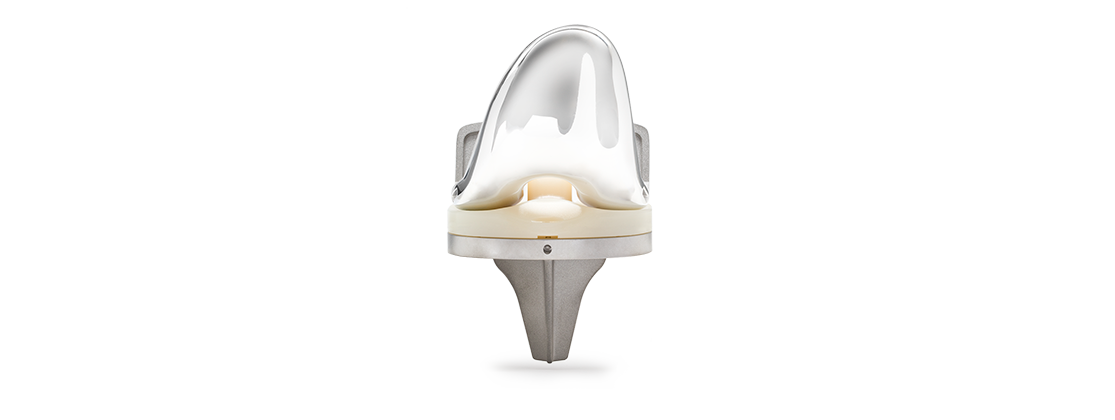
Ortho Development recently completed the first surgery using its Balanced Knee® System (BKS) in China at the Taiyuan Central Hospital in the Taiyuan, Shanxi province.
BKS implants and instrumentation address primary and revision procedures; more than 70,000 of the devices have been implanted worldwide. A patented locking mechanism is designed to minimize micro-motion between the plastic tibial insert and titanium tibial baseplates. Implants are offered in a range of sizes and options to allow anatomical fit for a variety of patients, the company says.
In July 2017, Ortho Development signed an exclusive regional distribution agreement with China Pioneer Pharma CPP for BKS. At the time, the first procedure was expected early this year; the slight delay appears to have arisen from steps necessary to gain regulatory approval.
Ortho Development’s product focus is total hip and knee replacement, along with trauma and spine devices. The company is privately held and its majority shareholder is Japan Medical Dynamic Marketing, a publicly-traded, Japanese medical device distribution company with offices in 15 major cities and a presence in over 2,000 hospitals throughout Japan.
JAV
Julie A. Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.




