
Content sponsored by Millstone Medical Outsourcing
Device companies and suppliers have made a considerable push to move from hospital-sterilized products to single-use, terminally sterilized devices. Increased infection rates at hospitals, regulatory implications on cleaning and the efforts of European and U.S. hospitals to cut costs have all driven sterilization changes in orthopaedic device packaging.
Here, we highlight more than a dozen announcements made in 2017 involving sterile-packed devices and products. Of note, as has been the trend in recent years, these announcements pertain to products in the spine or trauma space.
- DePuy Synthes introduced the Distal Radius Sterile Kit, a sterilized, single-use system of instruments and implants to treat wrist fracture.
- ECA Medical Instruments launched the sterile-packed, single-use Cervical-One instrument set, designed for one- and two-level cervical spine procedures.
- Flower Orthopedics launched new instruments in the Flower Efficiency (E-Kit) Advanced system; Flower Graft, a foot reconstruction platform with pre-shaped, bi-cortical allografts packaged in saline; and the addition of Headless Compression Screws to the Ready-for-Surgery cannulated screw set. The enhanced sterile E-Kits can reportedly replace the instruments in two sterilization trays.
- Gramercy Extremity Orthopedics received FDA 510(k) clearance to market the double sterile-packaged GEO Bone Screw, indicated for fractures, osteotomies, arthrodesis, osteochondritis and tendon reattachment.
- HD LifeSciences received FDA 510(k) clearance and commenced U.S. launch of the sterile-packed NanoHive, a system of interbody devices for ALIF, PLIF and TLIF procedures.
- In2Bones USA received FDA 510(k) clearance for its 5MS Fracture Repair and CoLag Locking Compression Screw systems, delivered sterile in individually packaged tubes.
- Meditech Spine received FDA 510(k) clearance for its CURE Lumbar Plating (LP) System, to be used along with the company’s lumbar Talos fusion devices. The CURE LP and Talos-A (HA) Lumbar IBF device will be combined, sterile and pre-packaged into one tray.
- Nextremity Solutions commenced limited launch of the disposable, single use, sterile-packed PiroVue Gastrocnemius Recession System instrument kit.
- OsteoMed released InstaFix, a shape memory fixation system delivered in a disposable, sterile kit.
- Paragon 28 launched JAWS, a Nitinol staple for use in osteotomy, arthrodesis and fragment fixation in the foot. JAWS is pre-loaded on a lightweight titanium inserter, provided in a sterile, disposable kit.
- SeaSpine commenced full commercial launch of the Vu aPOD Prime NanoMetalene System, a zero-profile, standalone anterior lumbar interbody device that can be configured to accommodate a patient’s pathology. Implants are delivered individually sterile-packaged.
- Tyber Medical received FDA 510(k) clearance for the TyFix all-in-one, sterile-packed extremity fixation system.
- WishBone Medical introduced the Exact Osteotomy System in single-used, sterile-packed kits.
- Xenco Medical announced U.S. expansion of the ASC CerviKit delivery and storage platform for its disposable Anterior Cervical Discectomy and Fusion systems. The ASC CerviKit, designed for ambulatory surgery centers, includes all of the company’s single-use cervical implant lines: pre-loaded interbodies and plates with single-use instruments locked together in a sterile package.
- XpandOrtho received FDA clearance for a sterile-packed, electronic soft tissue balancing instrument for total knee replacement.
- Zimmer Biomet commenced U.S. launch of the Avenue T TLIF Cage, featuring self-guided, curved and sterile-packed VerteBRIDGE plating for zero-profile, intra-discal fixation with a minimally invasive approach.
OEM Insight: Nextremity Solutions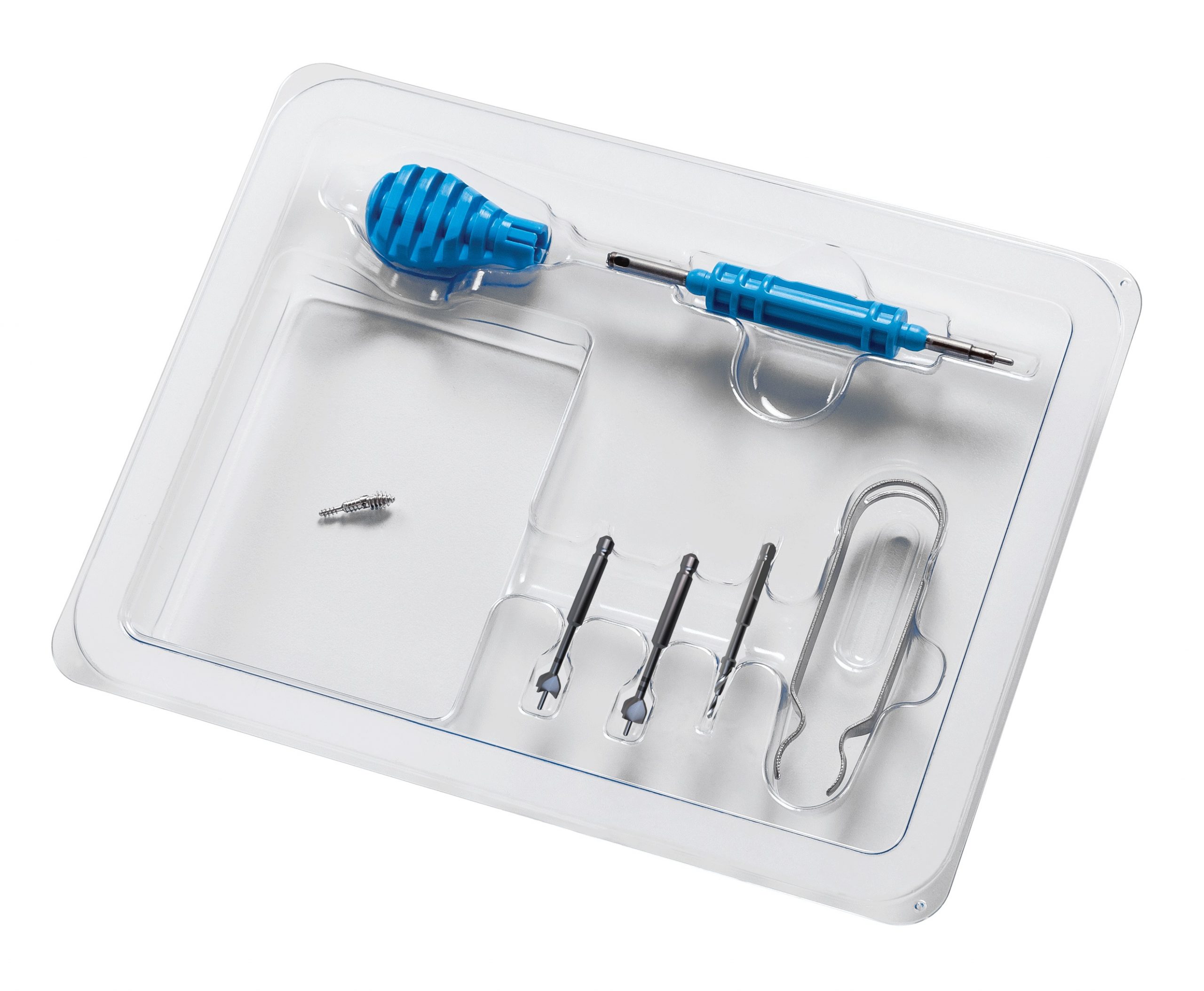

From the company’s beginnings, Nextremity Solutions’ surgeon founders had a vision for single-use, sterile-packaged, procedure-specific kits. This mandate challenged the company from materials and design perspectives in order to keep the cost of goods low and quality standards high, says Chief Commercial Officer Ryan D. Schlotterback.
Sterile packaging has offered numerous opportunities, including differentiation in the marketplace and efficiency from product launch, supply chain and sales perspectives due to the elimination of coordinating reusable kits in the field.
From a customer standpoint, Schlotterback says, “We’ve found that hospitals, as well as ambulatory surgical centers, find an advantage with these single-use, sterile packed kits as it saves them from reprocessing activities as well as having to inventory large amounts of reusable kits. This helps when getting product approval at the value analysis committee level. We’re committed to the single-use, sterile-packaged procedural kits as we move forward to develop new systems for foot and ankle, as well as other areas of musculoskeletal care. Our delivery systems continue to evolve to allow for more efficiency in the operating room, which will be important for future product launches, such as systems for Lapidus fixation, as well as a full foot plating system.”
Photo courtesy of Nextremity Solutions




