Share on:
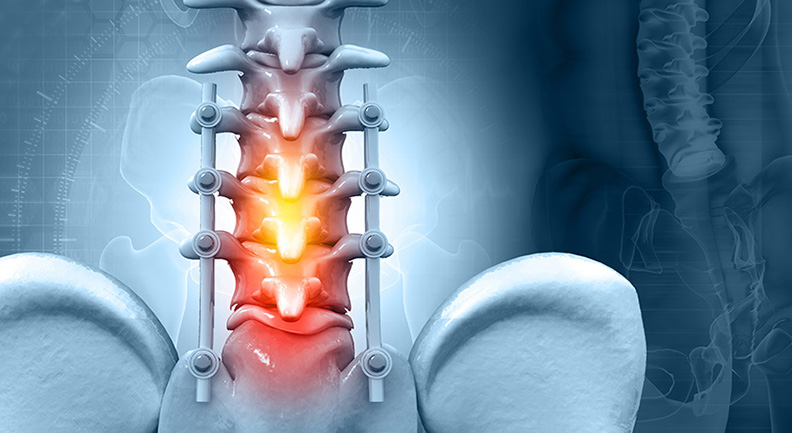
DSM entered into a partnership to supply Sayan Orthopaedics with Dyneema Purity® fiber for use in a trauma treatment device, comprising a combination of different types of Dyneema Purity® ultra-high-molecular-weight polyethylene (UHMWPE) fiber only. With a fiber 15 times stronger than steel, the braid device will be able to withstand high levels of force due to its strength and stiffness. Furthermore, the braid’s small diameter, high pliability and increased flexibility in combination with the device design, allows for easier adjustments during implantation. In addition to this the braid conforms to the shape of the bone, increasing the contact surface and lowering contact pressure. (Source: DSM)
RELATED ARTICLES
Janco Medical to Focus on Packaging and Cleanroom Manufacturing
Apr 10 2024 , Julie A. Vetalice
Evonik Grows Capacity for Customized RESOMER Powder Biomaterials
Apr 10 2024 , Julie A. Vetalice
Exploring Why Automation Leads to Improved Medical Device Manufacturing
Apr 09 2024 , Lowell
FDA De Novo for Orthobond’s Ostaguard Antibacterial Surface Treatment
Apr 08 2024 , Julie A. Vetalice